
Slowly dunk the LED into the liquid nitrogen, submerging completely. 
Attach the LED to the stick and hook the ends of the wires up from the LED to the power supply.This is why we see the orange light turn into colours that are higher on the electromagnetic spectrum when it is frozen in the liquid nitrogen. Since this gap is increased, when electrons in the conduction band fall to the valence band, they emit a higher energy light, meaning the light emitted has a shorter wavelength. When this happens, the bandgap in the semiconductors increases. When an LED is immersed in liquid nitrogen, the electrons lose thermal energy. Therefore, the size of this drop (the bangap), dictates what the wavelength of the emitted light. This is because as electrons fall from the conduction band to the valence band, they release photons of light energy. The bandgap is the difference in energy between the two bands, and this difference dictates the colour of the LED. These two bands are separated by what is known as a bandgap. This band is full of "holes" that electrons fall into as they pass from the conduction band of the cathode side to the val ence band of the anode side. The other side, called the anode side, has a valence band. This side has a conduction band which allows electrons to flow easily. This semiconductor is manufactured so that is has two sides. The most important part of the LED is the semiconductor material that the electrons flow through to create light. 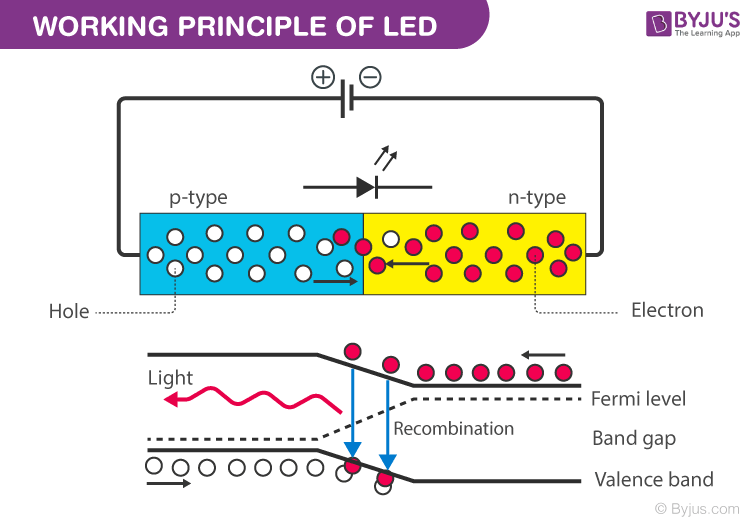
To understand the colour change, you first need to know a little something about how an LED works.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
